
Chemistry, 21.03.2020 10:33, odriskel49
The amounts of C and O are the same so the empirical formula has equal numbers of C and O atoms; that is, it has the general form CxHyOx. Determine the ratio of H/C (that is, y/x).

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, badgirl2005
This is a mixture that has the same composition throughout.
Answers: 1

Chemistry, 22.06.2019 16:30, ddmoorehouseov75lc
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 22:00, Porciabeauty6788
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 22.06.2019 23:10, carmenguabaoql9kv
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium. b)heavier than helium. c)the same weight as helium. d)dependent on the element that reacted with carbon.
Answers: 3
Do you know the correct answer?
The amounts of C and O are the same so the empirical formula has equal numbers of C and O atoms; tha...
Questions in other subjects:




Mathematics, 04.03.2020 04:00

Mathematics, 04.03.2020 04:00

Mathematics, 04.03.2020 04:00

Mathematics, 04.03.2020 04:00









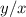 will be:
will be:





